Based on publicly available information from the Open EFSA and regulations issued by the European Commission, CIRS conducted a statistical analysis of the 2025 EU food additive submissions for corporate reference.
As of December 31, 2025, the approval status of EU food additives is shown in Figure 1.
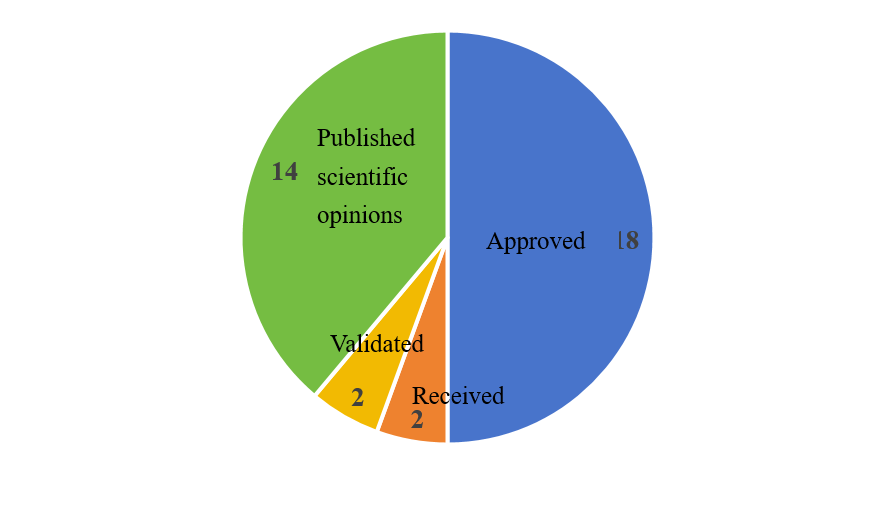
Figure 1. Approval status of EU food additives in 2025
EU Approved Food Additives
In 2025, the European Commission approved a total of 18 food additives, all of which were modification applications (see Table 1). According to EC No. 1331/2008 3(1), the food additives list may be updated either proactively by the EC or in response to applications from companies or Member States. Therefore, the application types include new food additives submissions, modification applications for already approved food additives, and re-evaluations of approved food additives.
In 2025, food additive approvals were dominated by modification applications. Compared with new additive approvals, the process for revising usage conditions or quality specifications of existing food additives may be more efficient. Regardless of the application type, careful review of the EU guidance when preparing dossier submissions is essential to ensure compliance and accelerate the approval process.
Table 1. List of food additives approved by the EC in 2025
No. | Code No. | Approved Date | Name of Substance | Application Type |
|---|---|---|---|---|
1 | (EU) 2025/651 | 2025.04.02 | Mono- and diglycerides of fatty acids (E 471) | Modification |
2 | Carnauba wax (E 903) | |||
3 | Lecithins (E 322) | |||
4 | Fatty acids (E 570) | |||
5 | (EU) 2025/652 | 2025.04.02 | Steviol glycosides produced by fermentation using Yarrowia lipolytica | Modification |
6 | (EU) 2025/666 | 2025.04.04 | Sodium carboxy methyl cellulose | Modification |
7 | Cellulose gum (E 466) | |||
8 | Cellulose (E 460) | |||
9 | Methyl cellulose (E 461) | |||
10 | Ethyl cellulose (E 462) | |||
11 | Hydroxypropyl cellulose (E 463) | |||
12 | Hydroxypropyl methyl cellulose (E 464) | |||
13 | Ethyl methyl cellulose (E 465) | |||
14 | Cross-linked sodium carboxy methyl cellulose | |||
15 | Cross linked cellulose gum (E 468) | |||
16 | Enzymatically hydrolysed carboxy methyl cellulose (E 469) | |||
17 | (EU) 2025/1150 | 2025.06.11 | Sodium ascorbate (E 301) | Modification |
18 | (EU) 2025/1337 | 2025.07.10 | Polyvinylpolypyrrolidone (E 1202) | Modification |
Food Additives Received and Validated by the EU
In 2025, the EFSA received and validated dossiers for two food additives each. Due to overlapping receipt and acceptance dates for monk fruit extract, a total of three food additives were involved (see Table 2).
Table 2. Food additives submitted and validated by EFSA in 2025
No. | Name of Substance | Application Type | Intended Function | Dossier Receipt Date | Acceptance Date |
|---|---|---|---|---|---|
1 | Tamarind seed polysaccharide | FA Application | Emulsifier, thickener, stabilizer | 2024.03.23 | 2025.05.27 |
2 | Monk fruit extract | FA Application | Sweetener | 2025.01.08 | 2025.06.23 |
3 | xylitol (E 967) obtained from a genetically modified Moniliella pollinis strain CD16243 | Modification | Sweetener | 2025.09.15 | / |
Food Additives Received Scientific Opinions
Among the 14 food additives for which EFSA issued scientific opinions in 2025, 3 new food additives applications, 2 modification applications, and 9 re-evaluations.
Table 3. List of food additives for which EFSA issued scientific opinions in 2025
Application Type | Name of Substance | Scientific Opinions |
|---|---|---|
FA Application | Jagua (genipin-glycine) blue | Intended function: colorant Safe under the proposed conditions of use. |
D-α-tocopheryl polyethylene glycol-1000 succinate | Intended function: emulsifier Safe under the proposed conditions of use. | |
Pea fibre concentrate (FIPEA) | Intended function: stabilizer, anti-caking agent, flour treatment agent, or foaming agent Safe under the proposed conditions of use. | |
Modification | Sorbitan monostearate | Safe under the proposed modified conditions of use. |
Rebaudioside M from stevia leaves | Safe under the proposed modified conditions of use. | |
Re-evaluation | Oxygen, Hydrogen, Neotame, Acesulfame-K, Pullulan, Butane, Isobutane, Propane, Mono- and diglycerides of fatty acids | Safe under the proposed conditions of use. |
Submissions from Chinese Companies
In 2025, the following Chinese companies submitted food additive dossiers to the EU, either accepted or having received scientific opinions.
Table 4. Chinese companies submitting food additives to the EU in 2025
No. | Name of Substance | Application Type | Application Status | Applicant |
|---|---|---|---|---|
1 | Monk fruit extract | FA Application | Received 2025-01-08 Accepted 2025-06-23 | Guilin Layn Ingredients Corp (China), SARAYA Co LTD; Tate & Lyle |
2 | Rebaudioside M from stevia leaves | FA Modification | Received scientific opinion-2025-05-14 Safe under the proposed conditions of use. | Ingia Bio |
CIRS Summary
Among the 2025 food additives submissions, apart from chemical substances, a significant proportion of submissions were plant-derived products, such as tamarind seed polysaccharide, monk fruit extract, pea fibre concentrate, and rebaudioside M from stevia leaves. Among these, sweetener submissions were prominent, reflecting the global sugar reduction trend, which has driven an increase in alternative sweetener applications to enrich consumer options.
Microbially-produced food additives represent a direction for modification applications, including steviol glycosides produced via Saccharomyces cerevisiae fermentation and rebaudioside M from stevia leaves (produced using genetically modified Pichia pastoris CGMCC 7539) for which scientific opinions were issued.
To meet the growing need for microbial strain safety assessment in synthetic biology, EFSA has published a new guidance on the safety assessment of microbial strains intended for the food chain, which provides more detailed scientific guidance, offering clear and efficient technical support for companies preparing food additives submissions.
If you need any assistance or have any questions, please get in touch with us via service@cirs-group.com.

