Definition
In Japan, food additives refer to substances used during food production or for the purpose of food processing, preservation, or improvement, including preservatives, sweeteners, colorants, flavorings, enzyme preparations, etc.
Regulatory Authorities
- Consumer Affairs Agency (CAA)
- Food Safety Commission of Japan (FSCJ)
Applicable Regulations
- Food Sanitation Act
- Guidelines for the Designation of Food Additives and Revision of Standards for Use
When is an Application Required?
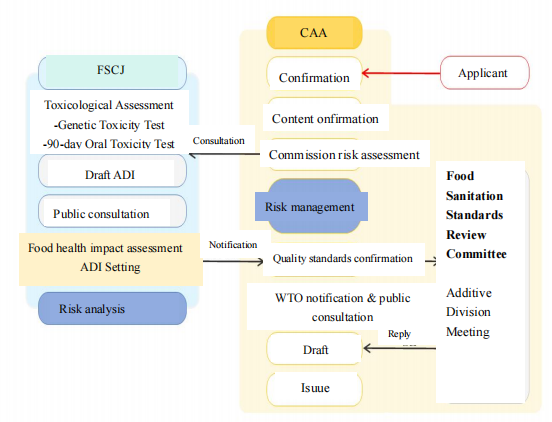
In Japan, only food additives approved under the Food Sanitation Act are permitted for use. An application must be submitted to the CAA when a new food additive is intended to be used in foods; or the scope of permitted foods for an existing food additive is proposed to be expanded. The application is reviewed by the FSCJ for safety evaluation. After the evaluation, the CAA makes the regulatory decision. The additive may be used in food only after the CAA promulgates the relevant regulation.
Note: Overseas companies must appoint a Japan-based responsible representative as the applicant.
Application Procedure for New Food Additives in Japan
Documentation Requirements
- Specification of ingredients
- Proposed standards for use
- Summary
- Name and intended use
- Source or discovery process
- Status of use in other countries
- Safety evaluations by international organizations
- Physicochemical properties and specifications
- Draft standards for use
- Efficacy data: ① Function as a food additive and comparison with similar additives ②Stability in food; ③ Impact on major nutrients in food
- Safety data
Application Timeline
From acceptance of the application to the promulgation of regulations by the CAA, the process typically takes about 18 months.
Food Additives Approved in the Past Five Years
New food additives: Ammonium bisulfite solution, Chitin glucan, Potassium DL-tartrate, Vinylimidazole–vinylpyrrolidone copolymer, Calcium phytate, Potassium bicarbonate, Polyvinyl alcohol
Food additives derived from GMMs (number of approvals): Asparaginase (1), Aminopeptidase (1), α-Amylase (19), α-Glucosidase (1), α-Glucosyltransferase (4), Exo-maltotetraohydrolase (2), Carboxypeptidase (1), Xylanase (5), Rennet (5), Glucoamylase (5), Glucose oxidase (3), Acid phosphatase (1), Cyclodextrin glucanotransferase (2), Terpene hydrocarbon (1), Allulose epimerase (1), Pullulanase (4), Protease (5), Pectinase (1), Hemicellulase (2), β-Amylase (1), β-Galactosidase (1), Phospholipase (8), Lipase (7), Riboflavin (2)
If you need any assistance or have any questions, please get in touch with us via service@cirs-group.com.