On Febuary 5, 2026, National Health Commission of the People’s Republic of China (NHC) issued an announcement (No. 1 of 2026) for “Three New Foods” with 22 products approved, including 5 new food raw materials, 11 new food additives, and 6 food-related products.
Details are as follows:
New food raw materials (5 types)
Gardenia oil
Name | Gardenia oil | |
Basic information | Source: fruit of Gardenia jasminoides J.Ellis | |
Brief introduction of production process | Made from the dried ripe fruit of Gardenia jasminoides as raw material, processed through pressing, extraction, degumming, deacidification, decolorization, deodorization, and dewaxing. | |
Specifications | Appearance | Light yellow to yellow oily liquid |
Fatty acid composition (percentage of total fatty acids): | ||
Oleic acid (C18:1), % | ≥ 20.0 | |
Linoleic acid (C18:2), % | ≥ 45.0 | |
Other information | The scope of use does not include infant and young children foods. Food safety indicators shall comply with the requirements for vegetable oils and fats as specified in the current Chinese National Food Safety Standards. | |
XiangYu peony flower
Name | XiangYu peony flower |
Basic information | Source: flowers of Paeonia suf ruticosa ‘Xiang Yu’ |
Other information | Not suitable for infants, pregnant women, and breastfeeding mothers. Labels and instructions must indicate unsuitable populations and consumption limits. Food safety indicators shall comply with the requirements for other vegetables as specified in the current Chinese National Food Safety Standards. |
Alginate oligosaccharide
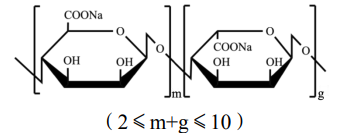
Name | Alginate oligosaccharide |
Basic information | Molecular formula:
|
Brief introduction of production process | Made from sodium alginate derived from brown algae such as kelp, sargassum, giant kelp, and bladder wrack, processed through hydrochloric acid dissolution, high-temperature degradation, sodium hydroxide neutralization, decolorization, purification, concentration, and drying. |
Recommended intake | ≤ 4 g/day (calculated based on a brown alginate oligosaccharide content of 90.0 g/100 g; products with higher content shall be converted according to the actual content). |
Other information | Not suitable for infants, pregnant women, and breastfeeding mothers. Labels and instructions must indicate unsuitable populations and consumption limits. Quality specifications and food safety indicators see Appendix. |
Bifidobacterium longum subsp. infantis YLGB-1496
Name | Bifidobacterium longum subsp. infantis LMG11588 |
Other information | The strain is approved and included in the List of Microorganisms That Can Be Used in Food. The food safety indicators shall comply with the requirements of the National Food Safety Standard for Microbial Preparations Used in Food Processing (GB 31639). Cronobacter spp. must not be detected (per 100 g). |
Camelina seed oil
Name | Camelina seed oil | |
Basic information | Camelina sativa (L.) Crantz | |
Brief introduction of production process | Made from Camelina sativa seeds as raw material, processed through screening, pressing, sedimentation, and filtration. | |
Specifications | Properties | Light yellow to brown yellow oily liquid |
Fatty acid composition (percentage of total fatty acids) | ||
Oleic acid (C18:1), % | ≥10.0 | |
Linoleic acid (C18:2), % | ≥13.0 | |
α-Linolenic acid (C18:3), % | ≥ 25.0 | |
Nervonic acid (C24:1), % | ≥ 0.5 | |
Erucic acid (C22:1), % | ≤ 4.5 | |
Other information |
| |
New food additives (11 types)
- New food enzymes
No. | Enzyme | Source | Donor |
1 | D-psicose 3-epimerase | Bacillus subtilis | Agrobacterium tumefaciensC58 |
2 | Glucose isomerase (xylose isomerase) | 168-N03 Bacillus subtilis 168-N03 | Streptomyces murinus |
3 | Deaminase | Streptomyces murinus | - |
4 | Sucrose 1-fructosyltransferase | Aspergillus japonicus | - |
The quality specifications for enzymes used in the food industry shall comply with the provisions outlined in the National Food Safety Standard for Food Additives – Enzymes for Food Industry (GB 1886.174).
- New food nutrition enhancer
3’-Sialyllactose sodium salt, 3’-SL
Food Cat. | Product Name | Usage Level | Note |
|---|---|---|---|
01.03.02 | Modified milk powder (limited to milk powder for children) | 0.06-0.28 g/L (Calculated on a pure substance basis and as consumed; for powdered products, the use level shall be converted according to the reconstitution ratio.) | When used in combination with 2’-Fucosyllactose (2’-FL), Lacto-N-neotetraose (LNnT), Galacto-oligosaccharides (GOS), Fructo-oligosaccharides (FOS), Polydextrose, and Raffinose, the total amount of these substances shall not exceed 64.5 g/kg. |
13.01.01 | Infant formula | ||
13.01.02 | Older infants and young children formula | ||
13.01.03 | Infant formula for special medical purposes |
Quality Specifications
This quality specification is applicable to the nutrition enhancer 3’-Sialyllactose sodium salt, produced using lactose and other materials as raw materials through fermentation, purification, and drying processes. The production strain used for manufacturing 3’-Sialyllactose sodium salt shall undergo a safety assessment and comply with the requirements specified in Appendix A.
- New food additives with expanded scope
No. | Name | Function | Cat. | Food Name | Maximum Level (g/kg) | Note |
1 | Carbon dioxide | Other | 14.01.02 | Drinking purified water | Used as needed for production | - |
14.01.03 | Other types of drinking water | |||||
2 | Gellan gum | Thickener | 01.05.01 | Light cream | Used as needed for production | - |
- Processing aids with expanded scope
No. | Name | Function | Application scope |
1 | bentonite | Clarifying agent | Starch syrup (limited to resistant dextrin) processing method |
- Food additives with supplementary quality specification
trans-Anethole
Function: food spice
Quality Specifications
This quality specification applies to the food additive Anethole, produced from propionic acid, acetic anhydride, and methyl benzoate as raw materials through chemical reaction.
2’-fucosyllactose,2’-FL
Application scope, usage level and specifications: The application scope and specification for 2'-Fucosyllactose shall follow the Announcement No. 8 of 2023 issued by the National Health Commission (except for the information on the production strains used for 2'-Fucosyllactose production in Appendix C). The information on the production strains for this new nutrition enhancer is provided in the table below.
Nutrition enhancer | Source | Donor |
2’-fucosyllactose | Escherichia coli BL21(DE3) | Escherichia coli |
Kluyveromyces lactis DSM70799 | Bacillus spp. |
Lacto-N-neotetraose
Function: food nutrition enhancer
Quality Specifications
The quality specifications for this substance shall be implemented in accordance with Announcement No. 8 of 2023 issued by the National Health Commission (except for the production strain information for Lacto-N-neotetraose listed in Appendix D). The production strain information for this new variety of nutritional fortifier is shown in the table below.
Nutrition enhancer | Source | Donor |
Lacto-N-neotetraose | Escherichia coli K-12 MG1655 | Neisseria spp. and Helicobacter spp. |
Escherichia coli BL21(DE3) | Neisseria meningitidis and Aggregatibacter kilianii |
New food-related products (6 types)
- Food contact additives with expanded use scope/level
Name | CAS number | Range of application | Maximum level |
Calcium sulfate | 7778-18-9 | PLA | 20 |
Erucamide; cis-13-Docosenamide | 112-84-5 | PVC | 2.7 |
- Food contact additives
Name | CAS number | Range of application | Maximum level |
Cobalt bis (2-ethylhexanoate) | 136-52-7 | UP | 0.02 |
- New food contact resin
Name | CAS number | Range of application | Maximum level |
Poly(ethylene 2,5-furandicarboxylate) | 28728-19-0 | Plastic | Use as needed in production |
Copolymer of ethyl acrylate, methacrylic acid and styrene | 25035-68-1 | Coatings and pants | 35 |
1,3-benzenedicarboxylic acid polymer with 1,4-benzenedicarboxylic acid, 1,4-cyclohexanedimethanol, decanedioic acid and 2,2-dimethyl-1,3-propanediol | 1345820-69-0 | Coatings and pants | 75 |
If you need any assistance or have any questions, please get in touch with us via service@cirs-group.com.