In 2025, U.S. GRAS notices remained highly active. To provide a comprehensive overview of the annual trends, CIRS Group conducted a systematic review and analysis of GRAS notices in 2025, offering insights and references for enterprises.
It should be noted that updates on the FDA website may be delayed, and many substances may have actually completed GRAS notices, but their status has not yet been updated. CIRS will closely monitor the FDA updates, and this discussion is based on the final information available on the FDA website for 2025.
Summary of FDA GRAS notices in 2025
The statistical results show that in 2025, a total of 91 substances had their information updated. Specifically, 30 substances were newly submitted during the year, 56 substances were newly filed, and 65 substances received new responses within the year.
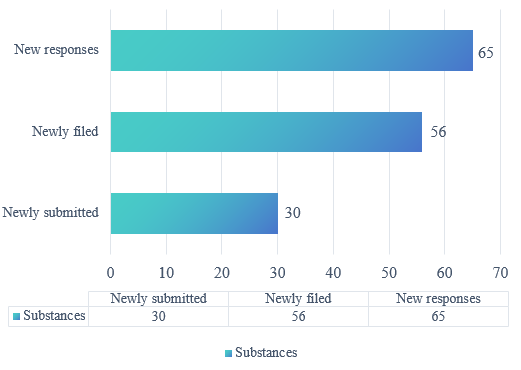
Figure 1. GRAS notice in 2025
In 2025, a total of 56 substances received “FDA has no questions” letters, seven ceased to be evaluated, and two notices did not provide a basis for a GRAS determination. Additionally, 26 substances are still pending.
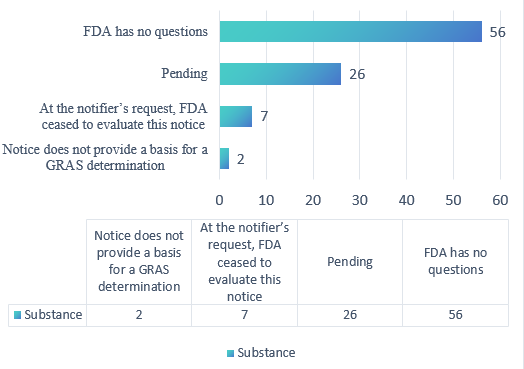
Figure 2. Overview of GRAS notices in 2025
Substances notified in 2025: a total of 56 substances
A total of 56 substances were notified in 2025, including 30 microorganism-related substances, three sugar substitute ingredients, three HMOs, and 20 substances in other categories.
The applicants originated from multiple countries, including the United States, China, Ireland, Germany, Denmark, Japan, France, the Netherlands, and India. Among them, U.S. and Chinese companies were the two main contributors of substances that obtained GRAS notices, with 25 substances and 19 substances, respectively.
Table 1. FDA GRAS substances notified in 2025
No. | GRN No. | Substance | Notified date | Enterprise |
|---|---|---|---|---|
1 | 1182 | Hydroxytyrosol | Feb 12, 2025 | Hangzhou Viablife Biotech Co, Ltd. (China) |
2 | 1187 | Polygalacturonase enzyme preparation produced by Trichoderma reesei expressing a gene encoding polygalacturonase from Aspergillus luchuensis mut kawachii | Feb 28, 2025 | AB Enzymes Inc. (US) |
3 | 1192 | Tannase enzyme preparation produced by Aspergillus oryzae | Mar 14, 2025 | Mitsubishi Chemical Corporation (Japan) |
4 | 1195 | Chymosin enzyme preparation produced by Aspergillus niger expressing a gene encoding chymosin from Erinaceus europaeus | Mar 6, 2025 | Chr. Hansen A/S (Denmark) |
5 | 1196 | Triacylglycerol lipase enzyme preparation produced by Trichoderma reesei expressing a gene encoding an engineered triacylglycerol lipase from Thermomyces lanuginosus | Mar 3, 2025 | AB Enzymes Inc. (US) |
6 | 1197 | Fructanase enzyme preparation produced by Trichoderma reesei AR-577 expressing the gene encoding fructanase from Lactobacillus crispatus | Mar 14, 2025 | AB Enzymes Inc. (US) |
7 | 1198 | Inositol | Feb 3, 2025 | Sichuan Bohaoda Biological Technology Co., Ltd. (China) |
8 | 1199 | Rhamnogalacturonan-I | Feb 28, 2025 | G3P Inc. (US) |
9 | 1200 | β-lactoglobulin produced by Komagataella phaffii strain VIPLA | Feb 28, 2025 | Vivici B.V. (the Netherlands) |
10 | 1201 | Lipase enzyme preparation produced by Komagataella phaffii expressing a gene encoding lipase from Yarrowia lipolytica | Mar 10, 2025 | Chr. Hansen A/S (Denmark) |
11 | 1202 | Soy leghemoglobin preparation from a strain of Komagataella phaffii | Mar 7, 2025 | Impossible Foods Inc (US) |
12 | 1203 | Rebaudioside M2 obtained by enzymatic treatment of steviol glycosides purified from the leaves of Stevia rebaudiana (Bertoni) Bertoni | Feb 12, 2025 | Guilin Layn Natural Ingredients Corp. (China) |
13 | 1204 | Lacticaseibacillus casei Zhang | Feb 19, 2025 | Beijing Scitop Bio-tech Co., LTD (China) |
14 | 1205 | Lactiplantibacillus plantarum DR7 | Mar 24, 2025 | Kaneka Americas Holding, Inc. (US) |
15 | 1206 | Rebaudioside M produced by enzymatic treatment of rebaudioside A purified from the leaves of Stevia rebaudiana (Bertoni) Bertoni | Feb 28, 2025 | Adorvia Biotechnology Co., Ltd. (China) |
16 | 1207 | Brazzein preparation produced by Aspergillus oryzae 90402 expressing a gene encoding for brazzein from Pentadiplandra brazzeana | Apr 10, 2025 | Nanjing Bestzyme Bio-Engineering Co. Ltd. (China) |
17 | 1208 | Lacto-N-triose II | Apr 29, 2025 | Shandong Henglu Biotechnology Co., Ltd. (China) |
18 | 1210 | Glucosyltransferase enzyme preparation produced by Bacillus subtilis expressing the gene encoding glucosyltransferase from Streptococcus salivarius | Aug 15, 2025 | Danisco US Inc. (US) |
19 | 1211 | Reduced minerals goat whey | Feb 4, 2025 | Crossway Foods Limited (Ireland) |
20 | 1212 | Liquid whole goat milk | Feb 7, 2025 | Crossway Foods Limited (Ireland) |
21 | 1213 | Goat milk lactose | Feb 21, 2025 | Crossway Foods Limited (Ireland) |
22 | 1214 | Bacillus clausii UBBC-07 spore preparation | May 7, 2025 | Unique Biotech Limited (India) |
23 | 1215 | Bacteriophage (phage) preparation specific to Listeria monocytogenes | Jun 6, 2025 | FINK Tec GmbH (Germany) |
24 | 1218 | Bacillus subtilis NRRL 68054 | Apr 29, 2025 | Microbial Discovery Group LLC (US) |
25 | 1219 | Recombinant bovine lactoferrin isolate produced by Komagataella phaffii M020 expressing the gene encoding bovine lactoferrin | May 7, 2025 | TurtleTree Inc (US) |
26 | 1221 | Heyndrickxia coagulans NRRL B-67744 | Jun 9, 2025 | Microbial Discovery Group, LLC (US) |
27 | 1222 | Saccharomyces cerevisiae OYR-542 | May 5, 2025 | Suntory Global Spirits (US) |
28 | 1223 | Sakura (Prunus serrulata Lindl.) flower distillate | Jun 3, 2025 | Suntory Global Spirits (US) |
29 | 1224 | Sakura (Prunus serrulata Lindl.) flower extract | Jun 3, 2025 | Suntory Global Spirits (US) |
30 | 1225 | Sakura (Prunus speciosa) leaf distillate | Jun 3, 2025 | Suntory Global Spirits (US) |
31 | 1226 | Orange (Citrus sinensis) extract | Jun 9, 2025 | BioActor B.V./Solabia Nutrition (Netherlands) |
32 | 1229 | Levilactobacillus brevis ATCC SD-7285 | Aug 15, 2025 | International Flavors & Fragrances, Inc. / Danisco USA, Inc. |
33 | 1230 | Vanillin preparation produced by Escherichia coli BL21(DE3) SI-VAN1 | Jun 11, 2025 | Sichuan Ingia Biosynthetic Co., Ltd. (China) |
34 | 1231 | Bacillus velezensis PTA-127359 | Jul 11, 2025 | BIO-CAT Microbials, LLC |
35 | 1232 | Lacticaseibacillus paracasei DSMZ 28047 | Aug 6, 2025 | SunWay Biotech Co., Ltd.(Taiwan) |
36 | 1233 | Sansho (Zanthoxylum piperitum) pepper distillate | Jul 8, 2025 | Suntory Global Spirits (US) |
37 | 1234 | Sansho (Zanthoxylum piperitum) pepper extract | Jul 14, 2025 | Suntory Global Spirits (US) |
38 | 1235 | Bifidobacterium lactis CCTCC M 2014588 | Sep 2, 2025 | Bioflag Co., Ltd (China) |
39 | 1236 | Algal oil (≥40% docosahexaenoic acid) from Aurantiochytrium limacinum H Sc-01 | Jun 10, 2025 | Shaanxi Healthful Bioengineering Co., Ltd. (China) |
40 | 1238 | 2'-fucosyllactose | Jun 17, 2025 | Cataya Bio (Shanghai) Co., Ltd. (China) |
41 | 1239 | Bifidobacterium animalis subsp. lactis PTA-126817 | Jun 11, 2025 | Wecare Probiotics Co., Ltd. (China) |
42 | 1240 | Lacticaseibacillus rhamnosus ATCC PTA-126815 | Jul 8, 2025 | Wecare Probiotics Co., Ltd. (China) |
43 | 1241 | β-lactoglobulin produced by Aspergillus oryzae | Sep 16, 2025 | Bon Vivant SAS (France) |
44 | 1242 | Short-chain fructooligosaccharides | Jul 14, 2025 | BENEO GmbH (Germany) |
45 | 1243 | Bacillus coagulans M2017813 spore preparation | Aug 21, 2025 | Thankcome Biological Science and Technology Co., Ltd. (China) |
46 | 1244 | Lactiplantibacillus plantarum DSM 21380 | Aug 5, 2025 | NORDWISE BioTech OÜ (US) |
47 | 1245 | Lactiplantibacillus plantarum DSM 21379 | Aug 5, 2025 | NORDWISE BioTech OÜ (US) |
48 | 1246 | Saccharomyces cerevisiae BY-1248 | Sep 22, 2025 | Berkeley Fermentation Science Inc. (US) |
49 | 1247 | β-Lactoglobulin produced by Kluyveromyces lactis CCTCC M20241460 | Sep 19, 2025 | Shanghai Changing Biotechnology Co., Ltd. (China) |
50 | 1248 | Yeast biomass produced by Kluyveromyces marxianus CCTCC M 20211265 | Sep 23, 2025 | Shanghai Changing Biotechnology Co., Ltd. (China) |
51 | 1249 | Ovalbumin produced by Trichoderma reesei ATCC 13631 | Sep 16, 2025 | Onego Bio, Inc. (US) |
52 | 1251 | Saccharomyces cerevisiae expressing a gene encoding a cysteine-thiol lyase from Staphylococcus hominis (S. cerevisiae "OYR-290") | Sep 23, 2025 | Omega Yeast Labs, LLC (US) |
53 | 1252 | Saccharomyces cerevisiae "BY-1568" | Sep 23, 2025 | Berkeley Fermentation Science Inc. (US) |
54 | 1264 | Lacticaseibacillus rhamnosus CGMCC18639 | Dec 9, 2025 | Beijing Scitop Bio-Tech Co., Ltd. (China) |
55 | 1265 | Saccharomyces cerevisiae "BY-1532" | Dec 11, 2025 | Berkeley Fermentation Science Inc. (US) |
56 | 1274 | 2'-fucosyllactose | Dec 8, 2025 | Tianjin Hesheng Biotechnology Co., Ltd. (China) |
Substances notified by Chinese companies in 2025: a total of 19 substances
In 2025, a total of 19 substances from Chinese companies received “FDA has no questions” letters, including eight microorganism-related substances, three sugar substitutes/sweeteners, three HMOs, and four substances in other categories.
Table 2. Substances notified by Chinese companies receiving “FDA has no questions” letters in 2025
No | GRN No. | Substance | Notified Date | Enterprise |
1 | 1264 | Lacticaseibacillus rhamnosus CGMCC18639 | Dec 9, 2025 | Beijing Scitop Bio-Tech Co., Ltd. (China) |
2 | 1274 | 2'-fucosyllactose | Dec 8, 2025 | Tianjin Hesheng Biotechnology Co., Ltd. (China) |
3 | 1182 | Hydroxytyrosol | Feb 12, 2025 | Hangzhou Viablife Biotech Co, Ltd. (China) |
4 | 1198 | Inositol | Feb 3, 2025 | Sichuan Bohaoda Biological Technology Co., Ltd. (China) |
5 | 1203 | Rebaudioside M2 obtained by enzymatic treatment of steviol glycosides purified from the leaves of Stevia rebaudiana (Bertoni) Bertoni | Feb 12, 2025 | Guilin Layn Natural Ingredients Corp. (China) |
6 | 1204 | Lacticaseibacillus casei Zhang | Feb 19, 2025 | Beijing Scitop Bio-tech Co., LTD (China) |
7 | 1206 | Rebaudioside M produced by enzymatic treatment of rebaudioside A purified from the leaves of Stevia rebaudiana (Bertoni) Bertoni | Feb 28, 2025 | Adorvia Biotechnology Co., Ltd. (China) |
8 | 1207 | Brazzein preparation produced by Aspergillus oryzae 90402 expressing a gene encoding for brazzein from Pentadiplandra brazzeana | Apr 10, 2025 | Nanjing Bestzyme Bio-Engineering Co. Ltd. (China) |
9 | 1208 | Lacto-N-triose II | Apr 29, 2025 | Shandong Henglu Biotechnology Co., Ltd. (China) |
10 | 1230 | Vanillin preparation produced by Escherichia coli BL21(DE3) SI-VAN1 | Jun 11, 2025 | Sichuan Ingia Biosynthetic Co., Ltd. (China) |
11 | 1232 | Lacticaseibacillus paracasei DSMZ 28047 | Aug 6, 2025 | SunWay Biotech Co., Ltd. (Taiwan) |
12 | 1235 | Bifidobacterium lactis CCTCC M 2014588 | Sep 2, 2025 | Bioflag Co., Ltd (China) |
13 | 1236 | Algal oil (≥40% docosahexaenoic acid) from Aurantiochytrium limacinum H Sc-01 | Jun 10, 2025 | Shaanxi Healthful Bioengineering Co., Ltd. (China) |
14 | 1238 | 2'-fucosyllactose | Jun 17, 2025 | Cataya Bio (Shanghai) Co., Ltd. (China) |
15 | 1239 | Bifidobacterium animalis subsp. lactis PTA-126817 | Jun 11, 2025 | Wecare Probiotics Co., Ltd. (China) |
16 | 1240 | Lacticaseibacillus rhamnosus ATCC PTA-126815 | Jul 8, 2025 | Wecare Probiotics Co., Ltd. (China) |
17 | 1243 | Bacillus coagulans M2017813 spore preparation | Aug 21, 2025 | Thankcome Biological Science and Technology Co., Ltd. (China) |
18 | 1247 | β-Lactoglobulin produced by Kluyveromyces lactis CCTCC M20241460 | Sep 19, 2025 | Shanghai Changing Biotechnology Co., Ltd. (China) |
19 | 1248 | Yeast biomass produced by Kluyveromyces marxianus CCTCC M 20211265 | Sep 23, 2025 | Shanghai Changing Biotechnology Co., Ltd. (China) |
Substances notified by Chinese companies in pending status in 2025
According to the latest information available on the FDA website, in 2025, Chinese companies had a total of 12 newly pending substances. HMOs were the primary category of submissions, with five substances pending, including 2′-fucosyllactose, lacto-N-neotetraose, lacto-N-tetraose, and 6′-sialyllactose sodium salt. There were four microorganism-related substances pending, including three microbial strains and one mycelial biomass. The remaining three substances were lycopene preparations, fungal oil, and L-ergothioneine.
Note: Due to potential delays in updates on the FDA website, it cannot be ruled out that some of these substances have already completed the review process but have not yet been publicly disclosed.
Table 3. Substances in pending status notified by Chinese enterprises in 2025
No. | GRN No. | Substance | Enterprise | |
|---|---|---|---|---|
1 | 1253 | Lycopene preparation produced by Saccharomyces cerevisiae JZL03 | Wuhan Hesheng Technology Co., Ltd. | |
2 | 1255 | Mycelial biomass of Fusarium compactum CGMCC 41312 (fungal protein) | MoreMeat (Guangzhou) Biotech Co., Ltd | |
3 | 1259 | Fungal oil (≥40% arachidonic acid (ARA)) from Mortierella alpina strain TKA-1 | ATK Biotech Co., Ltd. | |
4 | 1261 | 2'-fucosyllactose | Suzhou Yixi Biotech Co., Ltd. | |
5 | 1262 | 2'-fucosyllactose | Zhuhai Long Health Biotechnology Co., Ltd. | |
6 | 1270 | L-ergothioneine produced by Escherichia coli K-12 MG1655 expressing enzymes from Neurospora crassa and Mycolicibacterium smegmatis MC2 155 | Gene III Biotechnology Co., Ltd. | |
7 | 1272 | Lacto-N-neotetraose | Shenzhen Long Health Biotechnology Co., Ltd. | |
8 | 1273 | Lacto-N-tetraose | Shenzhen Long Health Biotechnology Co., Ltd. | |
9 | 1275 | 6′-sialyllactose sodium salt | Cataya Bio (Shanghai) Co., Ltd. | |
10 | 1277 | Autoclaved Parabacteroides goldsteinii “RV-01” | ReviveBio, Co. | |
11 | 1278 | Lactiplantibacillus plantarum CGMCC 6312 | Beijing Scitop Bio-Tech Co., Ltd. | |
12 | 1279 | Saccharomyces cerevisiae CCTCC M2025194 | Angel Yeast Co., Ltd. | |
Summary
The GRAS notice trends in 2025 show that microorganism-related substances – with 30 newly approved substances – were the undisputed mainstay of submissions, covering a wide range of probiotics, enzyme preparations, and emerging mycelial biomass products. Sugar substitutes and HMOs also maintained strong momentum in submissions, reflecting the market’s continued focus on sugar reduction and precision nutrition.
A key highlight of the year was the strong performance of Chinese biotechnology companies. Based on the latest information available from the FDA, 19 substances received “FDA has no questions” letters, while 12 additional substances remained pending, demonstrating the innovation capacity and growing competitiveness of Chinese companies in the global ingredients market.
*Data Source: The inventory of GRAS notices and the latest released accepted GRAS dossiers (updated as of 2025/12/30).
*Note:
1. As the FDA doesn’t disclose dossier acceptance dates, the statistics for substances in pending status are primarily based on the submission dates recorded in the released dossiers.
2. The statistics are based on the published dossiers with GRN No., and are for reference only.
About CIRS
The Food Division of CIRS Group was established in 2012 and it has a professional team specializing in US GRAS notices. The CIRS Food Division has extensive experience in various fields, covering GRAS, new food ingredients, new food additives, food contact materials, synthetic biology foods, EU Novel Foods, dietary supplements, and special dietary foods.
CIRS operates a fully-owned subsidiary in the US. By leveraging the expertise of the CIRS USA and the international team, it is able to provide enterprise various US food services, including but not limited to:
- US FDA GRAS Notices;
- US FDA Dietary Supplement Structure/Function Claim Notification;
- US New Dietary Ingredient (NDI) Notification;
- US FDA Registration of Food Facilities; and
- US Food Label/Advertisement Information Review.
If you need any assistance or have any questions, please get in touch with us via service@cirs-group.com.

